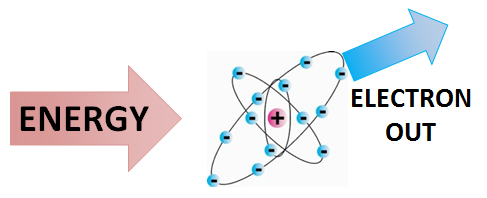
First Ionization Energy
IONIZATION FINDER
PERIODIC ATOMIC BINDING ENGINE
Atomic Pulse
The Energy Barrier. Ionization energy measures the Work Required to overcome the electrostatic pull of the nucleus.
- ⚛️ Ionization: The effective nuclear charge experienced by the valence electron.
- ⚡ Unit: Typically measured in **kJ/mol** or **electronvolts (eV)**.
- 📉 Radius Factor: Inversely proportional to the distance from the core.
Peak Ionization
Helium (He). Holding the record at 2372 kJ/mol, it represents the strongest electron-nucleus bond in nature.
- 🥇 Top Right: Helium sits at the extreme apex of periodic trends.
- 🛡️ Zero Shielding: No inner electrons to block the nuclear pull.
- 💎 Stability: A perfectly full 1s orbital creates immense resistance.
Reactive Peak
Francium (Fr). With an energy of only 393 kJ/mol, it sits at the absolute bottom of the energy barrier scale.
- 📉 Bottom Left: The trend follows the diagonal opposite of Helium.
- 🚀 Unstable: This low energy makes the atom highly prone to losing electrons.
- ⚛️ Shielding Max: Maximum electron layers lead to minimum nuclear pull.
Inert Logic
Atomic Stability. Noble gases possess Full Electron Shells, making them the most stable elements in the universe.
- 💎 Octet Rule: Having 8 valence electrons (except Helium's 2).
- 🌌 Monatomic: They do not naturally form molecules with themselves.
- 📉 Boiling Points: They have the lowest boiling points of any element group.
Metalloid Matrix
The Hybrid Zone. Metalloids are the Bridge between the rigid conductivity of metals and the insulating nature of nonmetals.
- 🔌 Semi-Conductive: Their electrical conductivity increases with temperature.
- 🔨 Brittle State: Unlike metals, they cannot be hammered into thin sheets.
- 🎨 Lustrous: Many, like Silicon, have a beautiful silver-grey sheen.
Solid Core
Structural Integrity. Metals provide the Conductive Foundation for both biological systems and modern infrastructure.
- 🏗️ Durability: High tensile strength for engineering.
- ⚡ E-Flow: High electron mobility for power grids.
- 🌡️ High Melting: Stable under extreme thermal stress.
Ionization Energy Paper
20 Questions: Values & Metal Identification
CERTIFICATE
CHEMISTRY MASTER
This certifies completion of the 20-Element Challenge
Score: 20/20
ELMO AI
Verification Office