Carbon Dating Caluculator
CARBON DATING
RADIOCARBON AGE ESTIMATOR
*Carbon-14 ($^{14}C$) is accurate up to 50,000 years.
YEARS OLD
Archaeological Horizon Reached
What is Carbon Dating?
Radiocarbon Dating is a chemical analysis used to determine the age of organic materials by measuring the amount of Carbon-14 remaining in a sample.
- ● The Isotope Clock: Every living thing absorbs carbon. When it dies, the intake stops, and the radioactive 14C begins to decay at a fixed rate.
- ● The Half-Life: 14C has a half-life of 5,730 years, meaning it takes that long for half of the atoms to disappear.
- ● Historical Anchor: This method allows archaeologists to date items back 50,000 years, providing a "Timeline of Life" for ancient civilizations.
Limit: ~50,000 Years
Chronological Limits
Every radioactive clock has a beginning and an end. In Carbon Dating, these are defined by the metabolic ceiling and the detection floor.
-
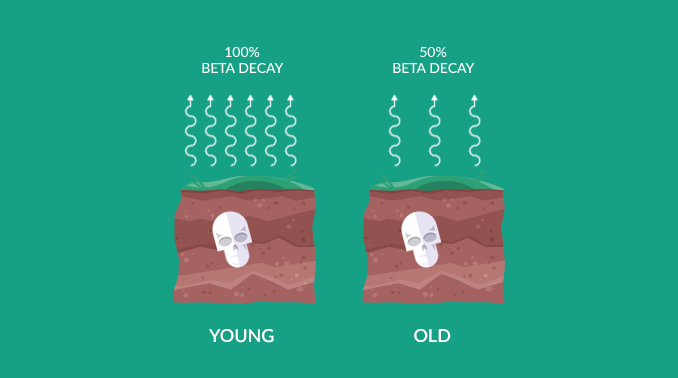
100%Organic Start: This is the "Zero Hour." While an organism is alive, it breathes or eats, keeping its 14C levels at 100% equilibrium with the atmosphere. Decay only begins the moment metabolism stops.
-
0.1%Decay Limit: After roughly 50,000 years (about 9 half-lives), the amount of remaining 14C becomes so small (<0.1\%) that it is indistinguishable from background radiation. At this point, the sample is "Radiocarbon Dead."
Samples older than the Decay Limit require Uranium-Lead or Potassium-Argon dating to measure deep geological time.
Isotope Concentration
Isotope Concentration (14-C) refers to the ratio of radioactive carbon remaining in a sample compared to the stable carbon-12. It is the primary variable in the decay equation.
-
RARelative Abundance: In nature, only one out of every trillion carbon atoms is 14-C. The concentration measures how many of those rare radioactive atoms haven't turned back into Nitrogen yet.
-
%Percentage Value: 100% concentration means the sample died "today." 50% concentration means exactly one half-life (5,730 years) has passed.
-
EQThe Equation: We calculate age using the natural log of the concentration:
t = ln(C) / -0.693 × 5730
% LEFT
System Parameters
Archaeological precision requires selecting the correct Isotope Source and accounting for Statistical Uncertainty.
ISOTOPE SOURCES (HALF-LIVES)
- • C-14 (5,730y): The modern Cambridge standard for radiocarbon dating.
- • Libby C-14 (5,568y): The original value used by Willard Libby (the "Libby half-life").
- • Tritium (12.3y): Used for dating groundwater and young organic liquids.
- • Ra-226 (1,600y): Radium-226, utilized for deep-sea corals and mineral deposits.
UNCERTAINTY (σ)
The Sigma (σ) value represents the confidence interval. Radiocarbon dating is never an absolute date, but a probability. A 1σ (Standard Deviation) means there is a 68% chance the actual age falls within the calculated range.
User Instructions
Follow these four steps to convert raw laboratory mass spectrometry data into a historical date.
Select Isotope: Choose your dating method. Use C-14 (5,730y) for modern archaeological standards or Libby for historical data comparison.
Input Concentration: Enter the percentage of 14-C remaining. Ensure this value is between 100% (Present) and 0.1% (Limit).
Set Uncertainty: Toggle the Sigma (σ) range provided by your lab report to define the margin of error.
Execute: The engine will process the natural log of your input to display the age in Years BP (Before Present).